Vascular differentiation and morphogenesis are spatially and temporally regulated by biochemical and biophysical cues of the ECM. Much of our understanding of these molecular regulations has been elucidated using in vivo systems or by developing well-defined in vitro culture models.
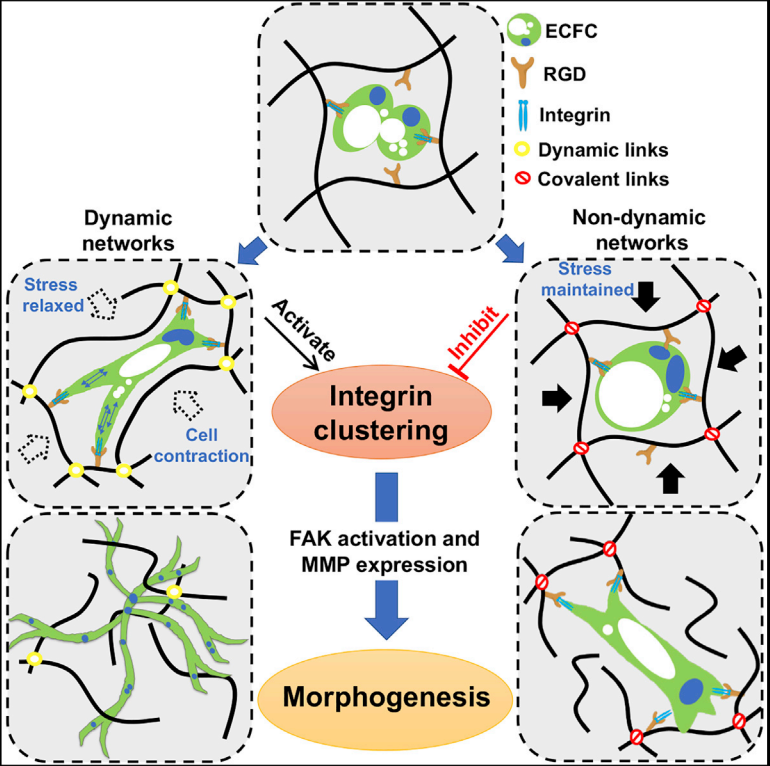
We develop and utilize synthetic, tunable materials to study various microenvironment aspects and their impact on vascular fate and assembly. In one set of studies, we addressed the need to activate cellular pathways to enable regenerative processes to proceed sequentially. Following deep burn wounding, we showed that amine-group functionalized on polymeric hydrogel accelerates inflammatory cell infiltration, leading to rapid degradation of the hydrogel scaffolds, promoting neovascularization that supported remarkable skin regeneration (1,2). For differentiation, we chemically modified surfaces to introduce various viscoelasticity properties. We found that substrate compliance guides EC fate by enhancing mesoderm induction through Wnt activation without adding small molecules (3). Using ECM micropatterning, we found that independent of chemical cues, spatially presented physical signals induce the self-organization of Brachyury-positive mesodermal cells in a RhoA/Rho-associated kinase (ROCK)-dependent manner (4). To elucidate 3D vascular morphogenesis, we engineered hydrogels that introduced both the adhesion and degradation motives into the polymer backbone. Vacuole and lumen formed through integrins α5β1 and αVβ¬3, while branching and sprouting were enabled by hydrogel degradation via MT1-MMP –activated MMP1, and MMP2 (5). We engineered a new biomaterial platform to study how matrix viscoelasticity regulates morphogenesis more recently. We use this platform to identify the viscoelasticity effect on vessel morphogenesis through cell contractility-integrin-FAK-MMP pathway activation (6). With these tunable biomaterials, we can decouple and uncover how microenvironmental cues regulate vascular fate and assembly and harness these processes towards therapeutics.
- Sun G, et al. Dextran hydrogel scaffolds enhance angiogenic response and promote complete skin regeneration during burn wound healing. Proc Natl Acad Sci U S A. 2011; 108:20976-20981.
- Shen Y-I, et al. Acellular hydrogels for regenerative burn wound healing: translation from a porcine model. J Invest Dermatol. 2015: 135:2519-2529
- Smith Q, et al. Compliant substratum guides endothelial commitment from human pluripotent stem cells. Sci Adv. 2017; 3: e1602883.
- Smith Q*, Rochman N*, et al. Cytoskeletal Tension Regulates Mesodermal Spatial Organization and Subsequent Vascular Fate. Proc Natl Acad Sci U S A. 2018; 115: 8167-8172.
- Hanjaya-Putra D, et al. Controlled activation of morphogenesis to generate a functional human microvasculature in a synthetic matrix. 2011;118:804-815.
- Wei Z*, Schnellmann R*, Pruitt HC, Gerecht S. Hydrogel network dynamics regulate vascular morphogenesis. Cell Stem Cell. 2020; 27:1-15.